We use cookies to ensure that we give you the best experience on our website. By using this website you agree to the Privacy Policy and Terms of Use.
Company Overview
About Tvardi
Tvardi Therapeutics is a clinical stage, biotechnology company developing a new class of breakthrough medicines for fibrosis-driven diseases. Founded in 2017 and based in Houston, Texas, the company is led by experienced entrepreneurs, innovative scientists, and dedicated physicians.
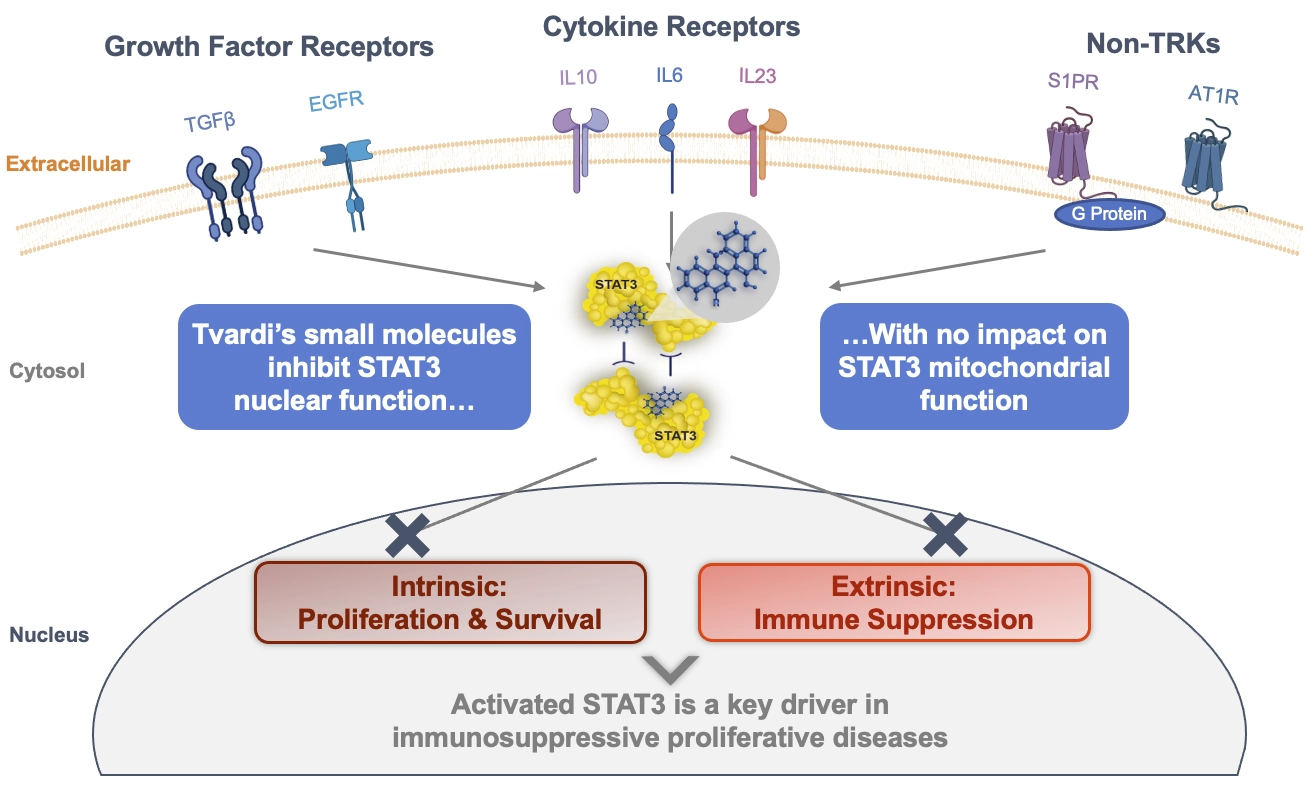
Persistent STAT3 activation can lead to uncontrolled chronic inflammation and fibrosis leading to a variety of chronic, debilitating diseases. STAT3 can be activated by a variety of cytokines, growth factors and non-tyrosine receptor kinases (non-TRKs), including IL-6 and TGF-β, which lead to pY-STAT3.
Tvardi is focused on the development of novel, oral small molecule therapies targeting STAT3 to treat fibrosis-driven diseases with significant unmet need. STAT3 is a central mediator across critical fibrotic signaling pathways that drive uncontrolled deposition, proliferation, survival, and immune suppression. STAT3 is also positioned at the intersection of many signaling pathways integral to the survival and immune evasion of cancer cells.
The company is conducting a Phase 2 clinical trial in hepatocellular carcinoma (NCT05440708) using its STAT3 inhibitor TTI-101. Separately, Tvardi’s second asset TTI-109, is an oral, small molecule STAT3 inhibitor that is structurally related to, yet chemically distinct from, TTI-101 and is designed to enhance our ability to target STAT3. Tvardi is currently conducting a Phase 1 trial of TTI-109 in healthy volunteers to evaluate safety, tolerability, and pharmacokinetics, as well as bioequivalence to TTI-101.
Collectively, the societal and economic impact of STAT3-dependent disease is significant. Tvardi is highly motivated to develop safe and effective small molecule inhibitors of STAT3 for use across numerous diseases.
Tvardi Therapeutics is a clinical-stage biopharmaceutical company developing a new class of breakthrough medicines for inflammatory and proliferative diseases.
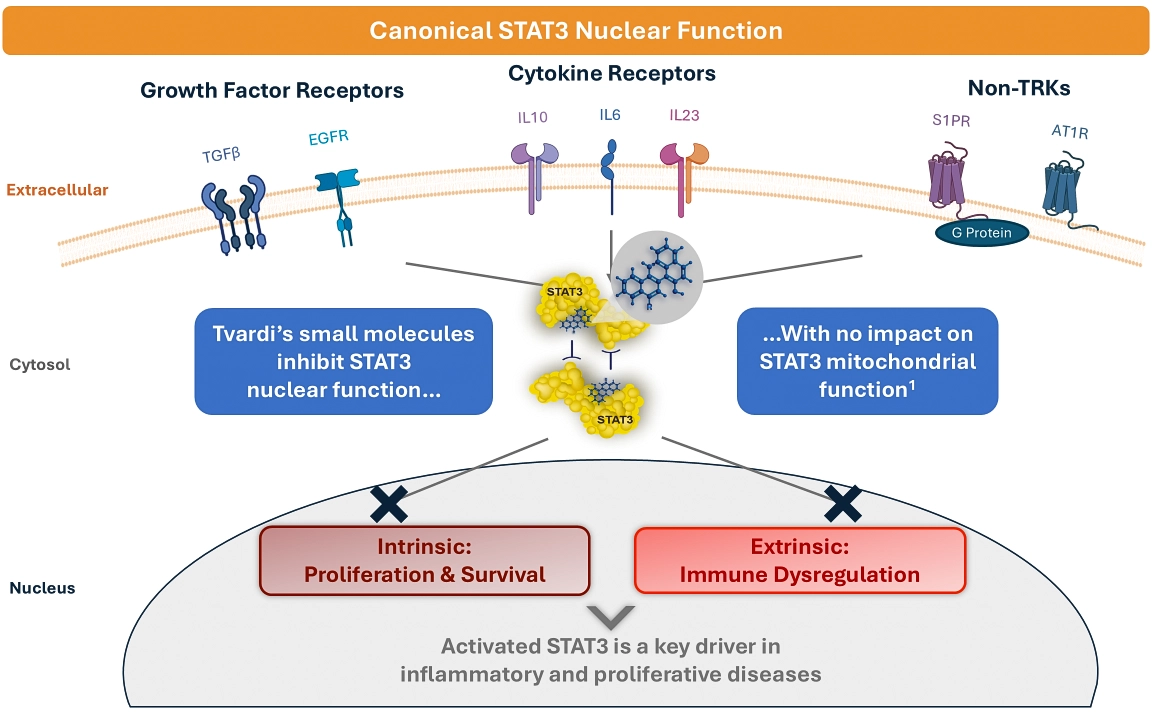
Tvardi is focused on the development of novel, oral small molecule therapies targeting STAT3 to treat inflammatory and proliferative diseases with significant unmet need. Based upon Tvardi’s founders’ seminal work and deep understanding of the transcription factor STAT3, the company has designed an innovative approach to directly inhibit STAT3, a highly validated, yet historically undruggable target. Leveraging this expertise, the company is developing a pipeline of STAT3 inhibitors with a differentiated mechanism of action and convenient oral dosing.
Tvardi’s pipeline includes two oral, small molecule STAT3 inhibitors: TTI-101 and TTI-109. TTI-101 is Tvardi’s first-generation direct STAT3 inhibitor, currently in a Phase 2 clinical trial in hepatocellular carcinoma (NCT05440708). TTI-109 is a phosphate prodrug of TTI-101 that is mechanistically identical to its parent molecule but is designed to enhance the company’s ability to target STAT3. Tvardi is currently conducting a Phase 1 trial of TTI-109 in healthy volunteers to evaluate safety, tolerability, and pharmacokinetics, as well as bioequivalence to TTI-101.
Tvardi believes their approach to directly inhibiting STAT3 enables them to develop product candidates with the potential to provide meaningful therapeutic benefit to patients with inflammatory and proliferative diseases.